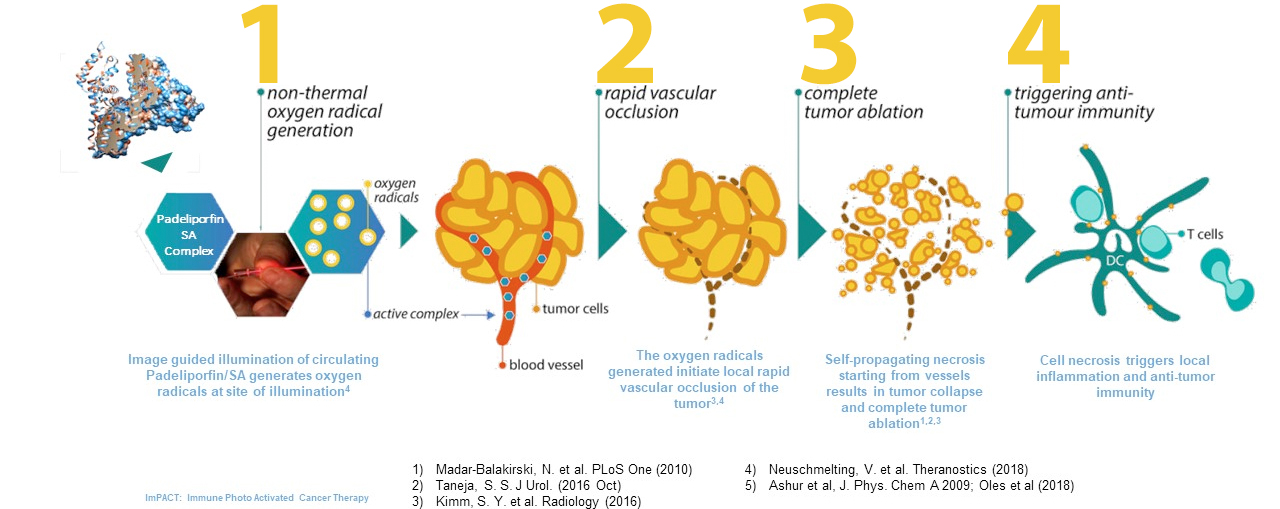
Padeliporfin Vascular Targeted Photodynamic (VTP) therapy is a novel Oncology technology platform that comprises the intravenous delivery of an inactive drug, Padeliporfin, which is then precisely activated only in the tumor micro-environment by non-thermal laser light delivered via optical fiber(s). This results in the immediate occlusion of the tumor blood supply followed by self-propagating immunogenic cell death and immune activation with minimal effect on surrounding healthy tissues.
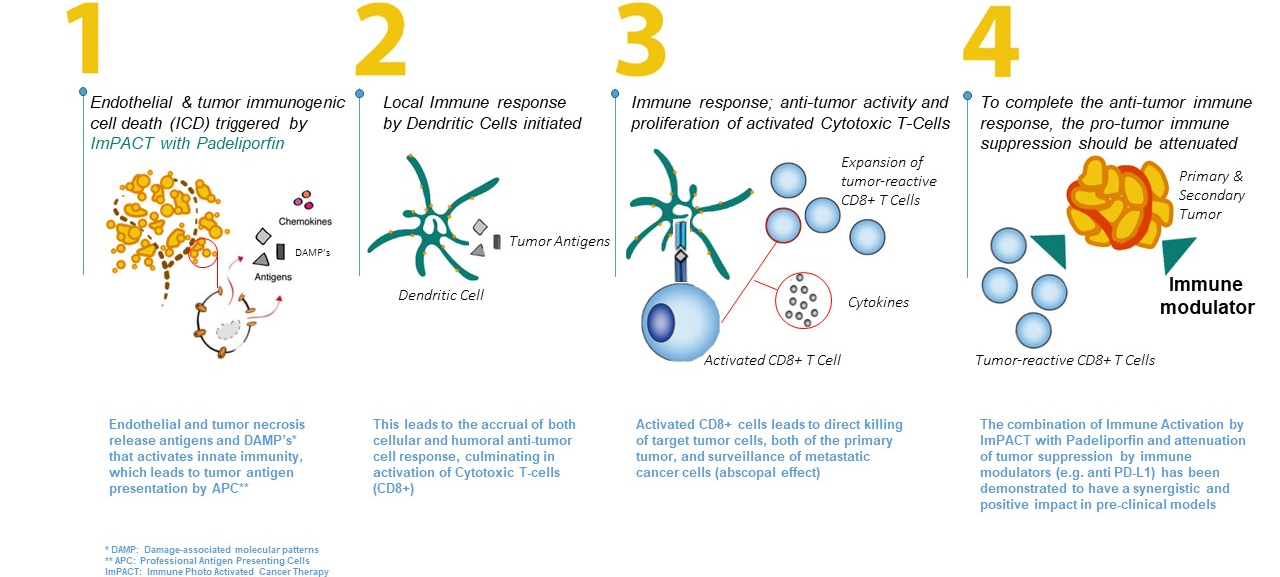
In pre-clinical studies, necrotic cell death has been demonstrated to activate an Immune response. The Padeliporfin VTP development program is exploring the potential to harness this immune response alone and in combination with various drugs including Anti-PDL1 and checkpoint inhibitors with the potential to regress remote tumors.
Padeliporfin VTP has the potential to address diseases where there is a high unmet medical need, especially where treatments are associated with significant morbidity that makes many patients ineligible for treatment. Strong safety and efficacy data previously obtained in localized prostate cancer studies demonstrates Padeliporfin VTP as a well-tolerated, convenient and efficacious treatment with the potential to become a mainstay cancer treatment approach for solid tumors at the localized and metastatic stage.